Articles
For information related to a specific standard, enter the standard number in the search bar below. If no result is returned, contact your account advisor for guidance.

Survey Ready? New Checklist Simplifies Record Preparation
Patient records are a vital part of the accreditation survey process. When a Surveyor arrives for your on-site survey, your organization must provide several patient record reports during the opening conference.
A new ACHC checklist is available to help you assess your organization’s compliance with patient record requirements.

CMS Publishes Final Rule Updating CLIA Regulations
Centers for Medicare & Medicaid Services (CMS) has published final rule CMS-3326-F to update Clinical Laboratory Improvement Amendments of 1988 (CLIA) regulations. Topics include the CLIA fee and alternative sanctions, along with histocompatibility and personnel requirements.

Personnel Background Checks Enhance Safety
Clients and patients depend on you to deliver excellent services in a secure environment. Performing background checks on personnel validates your commitment to providing safe, quality care.

Preparation Key to Effectively Handling Emergencies
ACHC Standards and Medicare Conditions for Coverage (CfCs) require organizations to develop an emergency preparedness plan.

CMS Changing CLIA Proficiency Testing Regulations
Staff and administrators of some clinical laboratories will have rules changes to keep up with in the months ahead as Clinical Laboratory Improvement Amendments (CLIA) proficiency testing regulations take effect.

Effective Date Set for Revised ABN Form
The revised Advance Beneficiary Notice of Noncoverage (ABN), Form CMS-R-131, is now available to download from the Centers for Medicare & Medicaid Services (CMS). Affected organizations must start using the updated ABN form by June 30, 2023.

Home Health: The Importance of Verifying Personnel Credentials
Recent federal arrests in a scheme to sell fake nursing diplomas serve as a critical reminder to home health agencies about the importance of verifying credentials for licensed personnel.

Changes to Know for Clinical Laboratories
COVID-19 tests and cholera testing remain current topics. The U.S. Food and Drug Administration has issued a bulletin about a COVID-19 test recall and has approved four more at-home antigen tests.
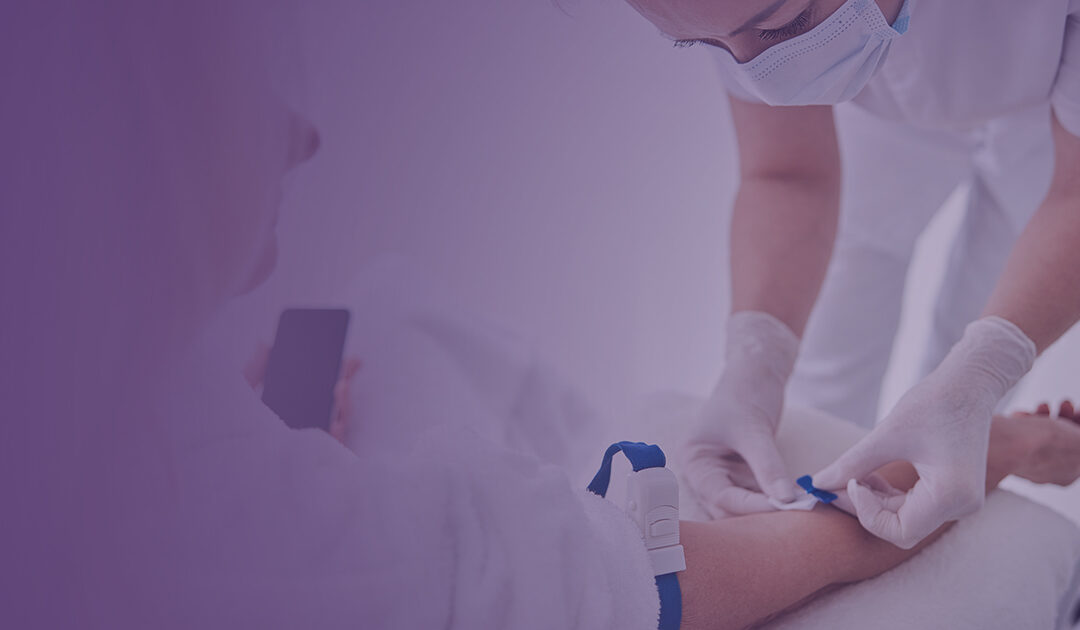
Home Infusion Therapy: Credential Verification Required for Licensed Personnel
Recent federal arrests in a scheme to sell fake nursing diplomas serve as a critical reminder to home infusion therapy organizations about the importance of verifying credentials for licensed personnel.

Demonstrating Separate Services for Home Infusion Therapy Accreditation
For home infusion therapy (HIT) suppliers, the Medicare Conditions for Coverage (CfCs) apply to an organization as a whole — including all clients/patients and not just Medicare beneficiaries.